News from Zentiva

November 25, 2020 / 2020
“Women shouldn’t be afraid of working in Science”, says Michaela Gajdošová, Dosage Forms Developer
What does the position of formulation manager mean and what is it like to develop dosage forms for a big pharmaceutical player? How does a young and promising scientist, whose work we will certainly hear about in the future, perceive the position of women in science? And how did the coronavirus pandemic affect her work? These are some of the few question we asked Michaela Gajdošová from Zentiva.
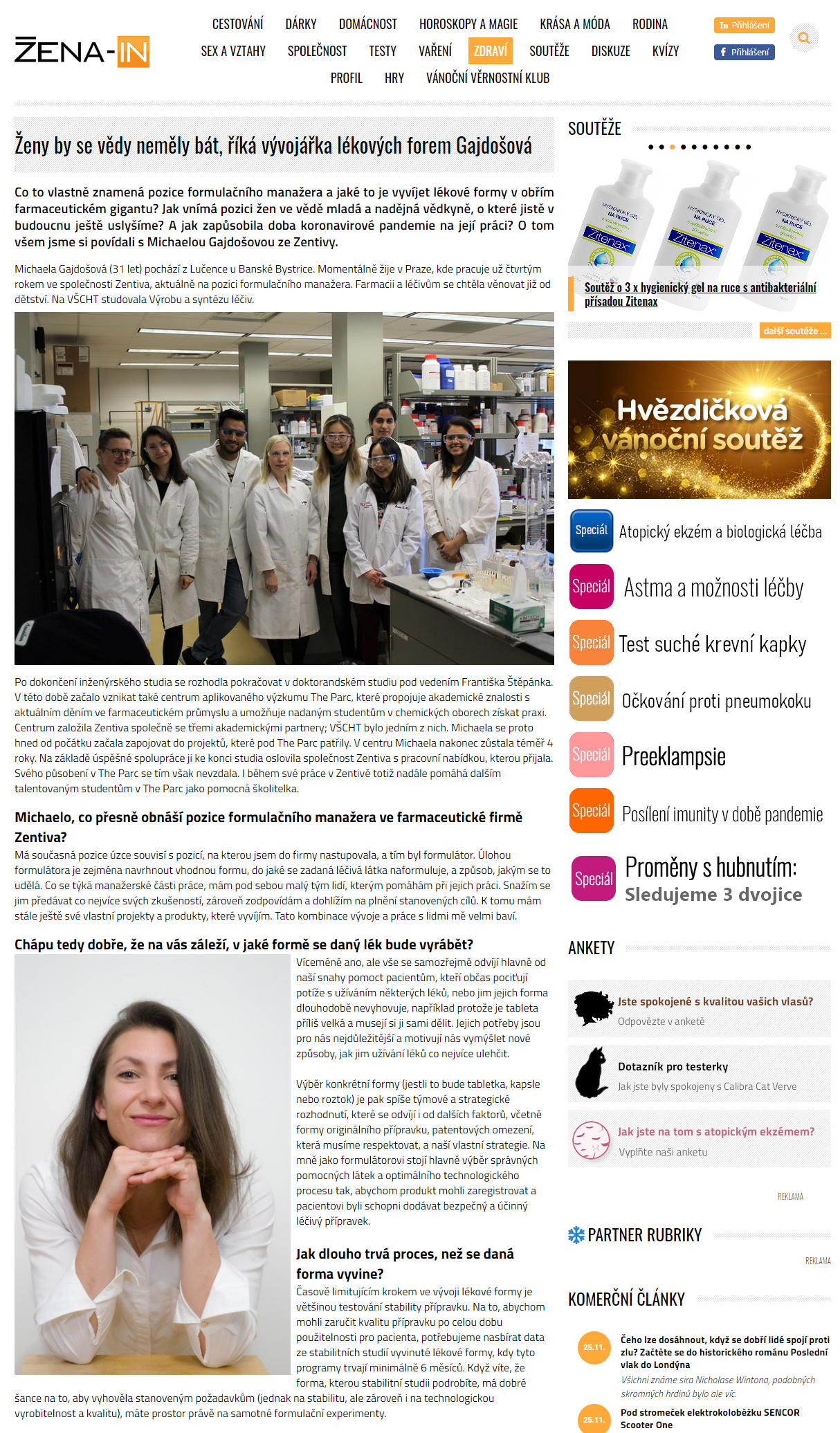
Michaela Gajdošová, 31, comes from Lučenec, Slovakia near Banská Bystrica. She currently lives in Prague, Czech Republic, where she has been working for Zentiva for the past four years as formulation manager. She wanted to work in pharmacy and medicines since her childhood. She studied the production and synthesis of drugs at the Institute of Chemical Technology (UCT).
After completing her engineering studies, she decided to continue her doctoral studies under the guidance of František Štěpánek, Scientific Director of The Parc. The Parc Applied Research Center, which bridges the gap between theoretical science and what happens in the real-life in pharma and helps talented students in the field of chemistry to gain hands-on experience, also began to emerge at this time. The Parc was founded by Zentiva together with three academic partners. UCT was one of them, so from the very beginning, Michaela got involved in projects that were part of The Parc pursued her career there for 4 years. Following a successful collaboration, she was approached by Zentiva at the end of her studies with a job offer. During her work at Zentiva, Michaela continues to work at The Parc as an assistant trainer.
Michaela, more precisely, what does the position of formulation manager at the pharmaceutical company Zentiva mean?
soThis position is closely related to the position I joined the company for at the beginning, and that is formulator. The role of a formulator is primarily to design appropriate dosage forms in which drug substances are formulated and to come up with a process to produce it. As for the managerial part of my job, I oversee a small team of people, whom I primarily help with their work. I try to pass them on as much of my experience as possible, at the same time I am responsible and supervising the fulfilment of the set goals. Besides, I still have projects and products of my own that I develop. I enjoy the combination of development and working with people.
So, we understand that it depends on you in what form the drug will be produced?
More or less yes, but, of course, it all depends primarily on patients who sometimes experience difficulties taking certain drugs or do not find some dosage forms suitable for a longer period, for example, because the tablet is too big and they have to divide it in half by themselves. Their needs are most important to us and motivate us to come up with new ways to make taking medication as easy and safe as possible for them.
When it comes to the choice of a particular form (whether it is a tablet, capsule, or solution), it is more of a team and strategic decision, which depends also on other things, such as the form of the original product, patent restrictions that we must respect, and our strategy. As a formulator, it is mainly up to me to choose the right excipients and the optimal technological process so that we can register the product and be able to repeatedly deliver a safe and effective medicine to the patient.
How long does the development process take?
The time-limiting step in the development of the dosage form is mostly testing the stability of the preparation. To guarantee the quality of the product for the entire period of use for the patient, we need to collect data from the stability studies of the developed dosage form, which last for at least 6 months. If you know that the form you submit to the stability study has a good chance of meeting the set requirements (both for stability, but also for technological manufacturability and quality), you have room for formulation experiments.
What are the most popular dosage forms with Czech patients?
I do not have any statistics available, but when developing a dosage form, we always take into account what would suit the patient best in the given case. For some products, it is necessary to accept the form that is on the market, because it has proven to be the most effective and this is always the most important factor. For preparations, where this is only slightly possible, we always consider how to add added value for the patient. And this may concern the method and frequency of administration, the reduction of side effects, or the expansion of the available strengths of the drug on the market, which eliminates, for example, the division of the tablet in half as mentioned above. From personal experience, it is most pleasant for me if the medicine can be taken comfortably, the tablet or capsule is not too big, does not have an unpleasant taste and I can take the medicine for a longer time. I think that solid dosage forms such as tablets and capsules are still the most common and common forms. At Zentiva, we are also working on other, innovative dosage forms that should be available in our portfolio in the future and will bring many benefits to patients.
What are your memories of studying at The Parc?
I have very nice memories. I got into a team of people who inspired me. Whether they were students, supervisors at school, or people in Zentiva. Professor Štěpánek had an incredibly positive and creative approach to scientific activities, and at the same time, he was able to support his students in participating in international conferences or collaborations, which was a very valuable experience for me. The opportunity to present and consult my research and results at Zentiva with experienced professionals in the field has given me a completely different view of research, what I do, and how it can be useful. I had the opportunity to see how it works on both sides, in academia and at the same time in the industry. The academy and the corporation are two different worlds that can complement each other. I was thrilled by this concept and so far, it has been very beneficial to me. In The Parc, you can learn something extra about traditional academic research. In addition to it, there is a great atmosphere and the tutors have a real interest in the students’ development, not only professionally but also personally. Creative team building was a matter of course.
To whom would you recommend doctoral studies at The Parc?
I would recommend it to anyone considering a career in pharmaceutical development, whether on the company side or in the academic field. If I were an academic, I would like to direct my research so that it has a real and positive impact on the patient. Besides, the real experience of how pharmaceutical companies and this industry work is very good. At The Parc, students come into contact with all the work and projects at joint conferences and thus have an overview of what is happening there. A program is also being prepared that will help students understand the principles of development in a pharmaceutical company, where it takes place and what it all entails. If you are considering working in the industry, studying at The Parc is an ideal opportunity to gather experience and present yourself as a capable potential employee.
In your opinion, are there enough women involved in science or do men predominate?
Paradoxically, there was a large predominance of women in my field during my studies at the Institute of Chemical Technology, specifically in the production and synthesis of drugs. And by that, I mean really big predominance. At Zentiva, I was very pleasantly surprised by the policy towards young women. Not only have they been openly interested in me as a woman, but overall, Zentiva as a company gives young women in science the opportunity and flexibility to organize work if you decide to start a family as a woman.
However, according to my information, global statistics still show that there are fewer women in science. Despite the numbers, I must say that I have many women around who inspire me with their approach and scientific work – either students and academic staff.
There are many influential women in science in general. I think they shouldn't be afraid of working in the field. Moreover, the feminine element always brings something new and different for each work. I believe that in time the ratio of men and women in science will equalize.
What moment in your career do you value the most?
I can't currently choose one particular moment. To name some, the opportunity to be a part of The Parc, the opportunity in the team of formulators, and finally my superior's confidence that I would manage the position of team leader, led me in my career to where I am currently, and I am personally very satisfied with it. I was also sincerely pleased when I ended my academic journey by successfully defending my Ph.D., even though it may sound trivial. I would also like to say that I was very happy to see the MRI (magnetic resonance imaging) image from the tablet, which I took in collaboration with a colleague on her research, on the cover of Pharmaceutical Research 2017.
What are you currently working on?
I am currently working on several preparation projects, exceptionally dealing with the development of liquid dosage forms or ampoules with infusion solutions, which is a special feature for us of the common development of products in a solid dosage form. I also work as a tutor for several students at The Parc, working on 3D printing of dosage forms, the use of powder rheometers to streamline the development of solid dosage forms, or the implementation of mathematical modelling to optimize and scale technological operations. I am also constantly working to make our teamwork efficient and to work well together. It is also worth mentioning the effort to map and optimize the work with information flows in the development - specifically, the creation of a single system for sharing all documents.
How did this year's coronavirus pandemic affect your work?
Have there been any changes, restrictions?
I must say that Zentiva has approached this situation very responsibly. It has put in place a series of internal measures that we all follow because we know they provide us with safety at work. These include keeping a safe distance, wearing masks and respirators, and using disinfectant regularly. As part of measures to protect production, the possibility of working from home was, of course, introduced. However, when you need to be physically present in the laboratory to develop a product, such a recommendation is hard to follow. Therefore, we meet only in one department and wear masks or respirators. Traditional meetings do not take place at all, everything goes through remote meetings online, whether you are at home or in the company. At the same time, I came across interesting projects, such as the development of disinfectants Sanicor, and learned what requirements these products must meet when being introduced to the market.